This post has been read 1507 times!
 May 2, 2023- by Steven E. Greer, MD
May 2, 2023- by Steven E. Greer, MD
A new medical meeting poster shows the results of a double blind, randomized, controlled trial of ivermectin as a therapy for COVID. The drug works very well.
MedinCell is the drug company that conducted the trial. They are making a long-acting version of ivermectin to sell at higher prices than the generic version.
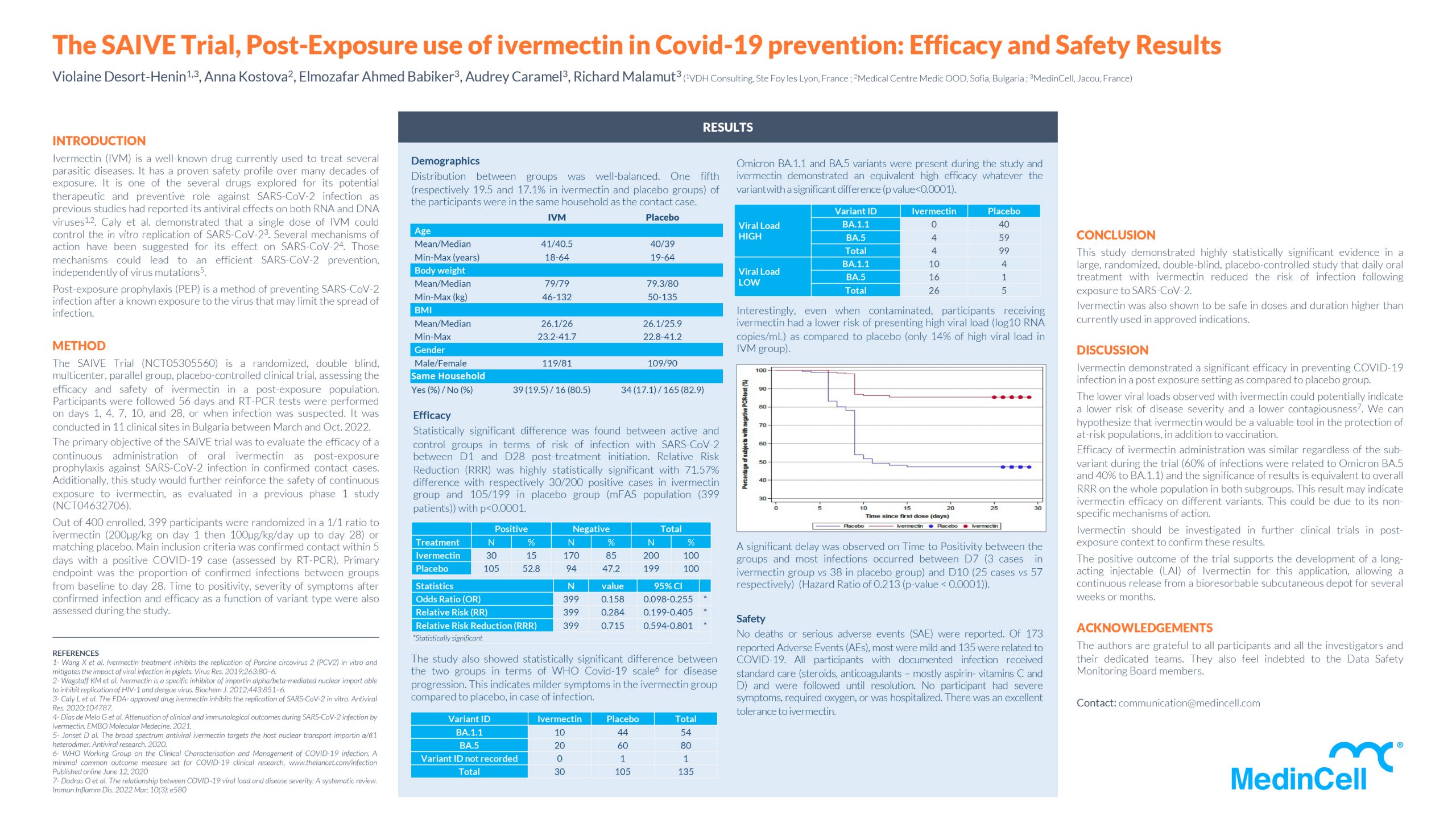
METHOD
The SAIVE Trial (NCT05305560) is a randomized, double blind, multicenter, parallel group, placebo-controlled clinical trial, assessing the efficacy and safety of ivermectin in a post-exposure population.
Out of 400 enrolled, 399 participants were randomized in a 1/1 ratio to ivermectin (200μg/kg on day 1 then 100μg/kg/day up to day 28) or matching placebo. Main inclusion criteria was confirmed contact within 5 days with a positive COVID-19 case (assessed by RT-PCR). Primary endpoint was the proportion of confirmed infections between groups from baseline to day 28. Time to positivity, severity of symptoms after confirmed infection and efficacy as a function of variant type were also assessed during the study.
Efficacy
Statistically significant difference was found between active and control groups in terms of risk of infection with SARS–CoV–2 between D1 and D28 post–treatment initiation. Relative Risk Reduction (RRR) was highly statistically significant with 71.57% difference with respectively 30/200 positive cases in ivermectin group and 105/199 in placebo group (mFAS population (399 patients)) with p<0.0001
The study also showed statistically significant difference between the two groups in terms of WHO Covid-19 scale6 for disease progression. This indicates milder symptoms in the ivermectin group compared to placebo, in case of infection.
Omicron BA.1.1 and BA.5 variants were present during the study and ivermectin demonstrated an equivalent high efficacy whatever the variant with a significant difference (p value<0.0001).
Interestingly, even when contaminated, participants receiving ivermectin had a lower risk of presenting high viral load (log10 RNA copies/mL) as compared to placebo (only 14% of high viral load in IVMgroup).
Safety
No deaths or serious adverse events (SAE) were reported. Of 173 reported Adverse Events (AEs), most were mild and 135 were related to COVID-19. All participants with documented infection received standard care (steroids, anticoagulants – mostly aspirin- vitamins C and D) and were followed until resolution. No participant had severe symptoms, required oxygen, or was hospitalized. There was an excellent tolerance to ivermectin.
DISCUSSION
Ivermectin demonstrated a significant efficacy in preventing COVID-19 infection in a post exposure setting as compared to placebo group. The lower viral loads observed with ivermectin could potentially indicate a lower risk of disease severity and a lower contagiousness. We can hypothesize that ivermectin would be a valuable tool in the protection of at-risk populations, in addition to vaccination.
Efficacy of ivermectin administration was similar regardless of the subvariant
during the trial (60% of infections were related to Omicron BA.5 and 40% to BA.1.1) and the significance of results is equivalent to overall RRR on the whole population in both subgroups. This result may indicate ivermectin efficacy on different variants. This could be due to its nonspecific mechanisms of action.
Ivermectin should be investigated in further clinical trials in postexposure context to confirm these results.
The positive outcome of the trial supports the development of a longacting injectable (LAI) of Ivermectin for this application, allowing a continuous release from a bioresorbable subcutaneous depot for several weeks or months.

Thank you for this VERY informative piece, as the MSM will never print it.